| Citation: |
Zhizai Li, Zhiwen Jin. HI hydrolysis-derived intermediate as booster for CsPbI3 perovskite: from crystal structure, film fabrication to device performance[J]. Journal of Semiconductors, 2020, 41(5): 051202. doi: 10.1088/1674-4926/41/5/051202
****
Z Z Li, Z W Jin, HI hydrolysis-derived intermediate as booster for CsPbI3 perovskite: from crystal structure, film fabrication to device performance[J]. J. Semicond., 2020, 41(5): 051202. doi: 10.1088/1674-4926/41/5/051202.
|
HI hydrolysis-derived intermediate as booster for CsPbI3 perovskite: from crystal structure, film fabrication to device performance
DOI: 10.1088/1674-4926/41/5/051202
More Information
-
Abstract
Nowadays, inorganic CsPbI3 perovskite solar cells (PSCs) have become one of the most attractive research hotspots in photovoltaic field for its superior chemical stability and excellent photo-electronic properties. Since the first independent report in 2015, the power conversion efficiency (PCE) of CsPbI3 based PSCs has sharply increased from 3.9% to 19.03%. Importantly, during the developing process of CsPbI3 PSCs, HI hydrolysis-derived intermediate plays an important role: from stabilizing the crystal structure, optimizing the fabricated film to boosting the device performance. In this review, the different crystal and electronic structures of CsPbI3 are introduced. We then trace the history and disputes of HI hydrolysis-derived intermediate to make this review more logical. Meanwhile, we highlight the functions of HI hydrolysis-derived intermediate, and systematically summarize the advanced works on CsPbI3 PSCs. Finally, the bottlenecks and prospects are revealed to further increase the CsPbI3 PSCs performance.-
Keywords:
- CsPbI3,
- HI,
- intermediate,
- crystal structure,
- stability
-
References
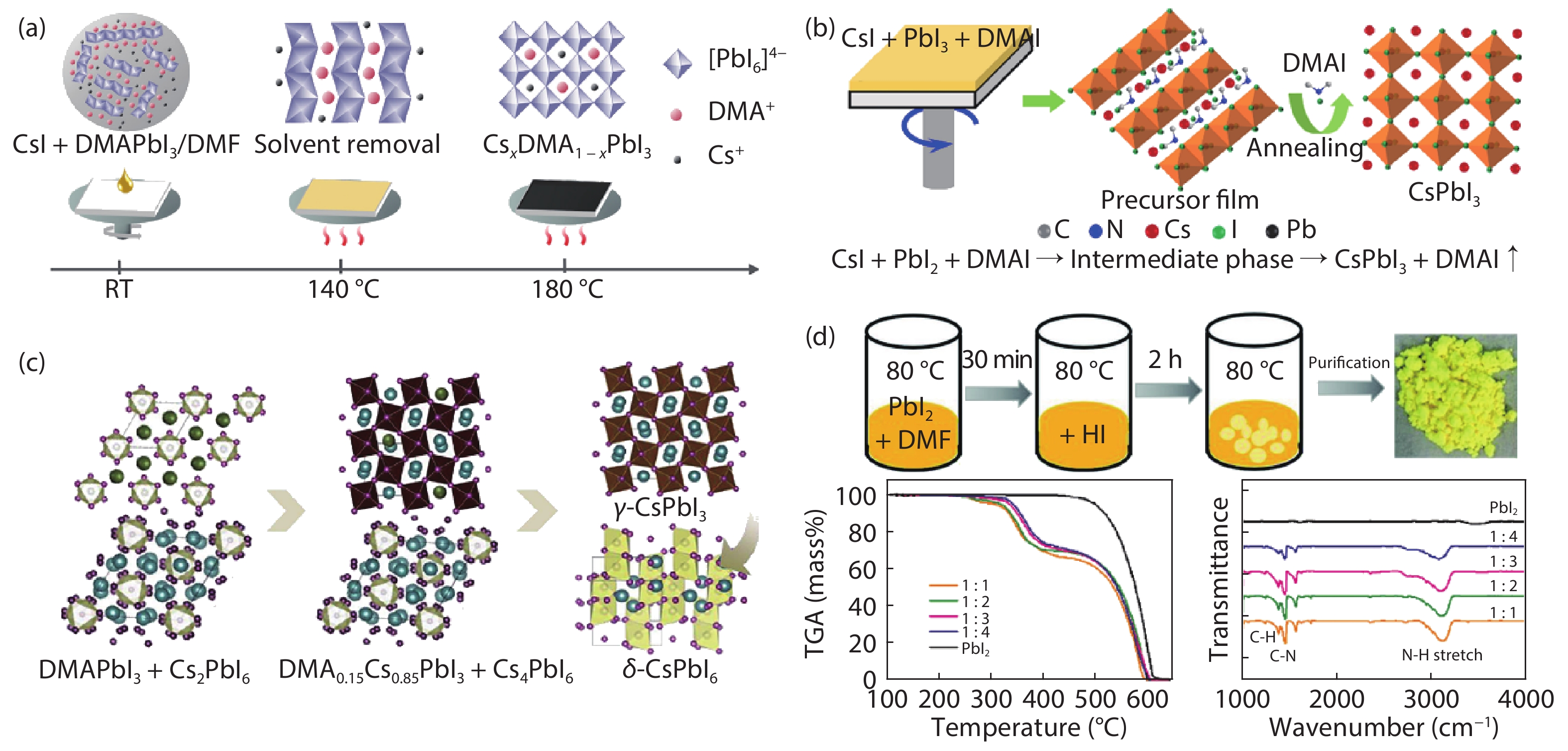
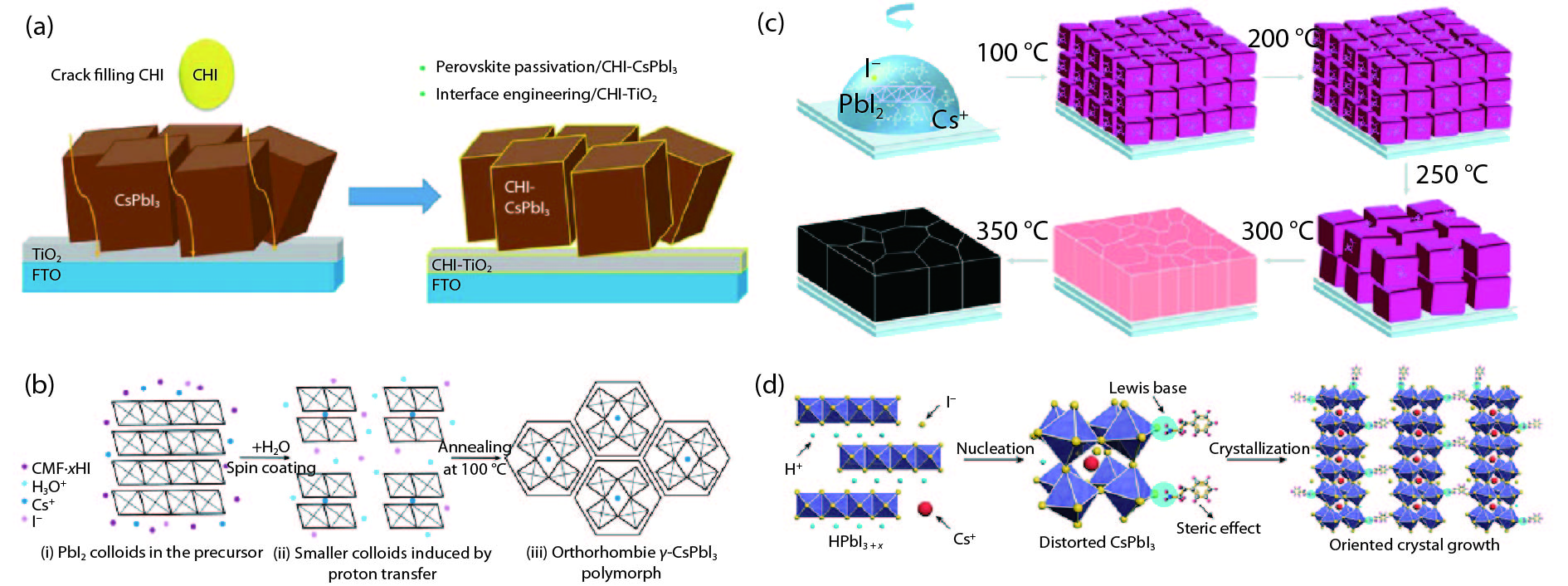
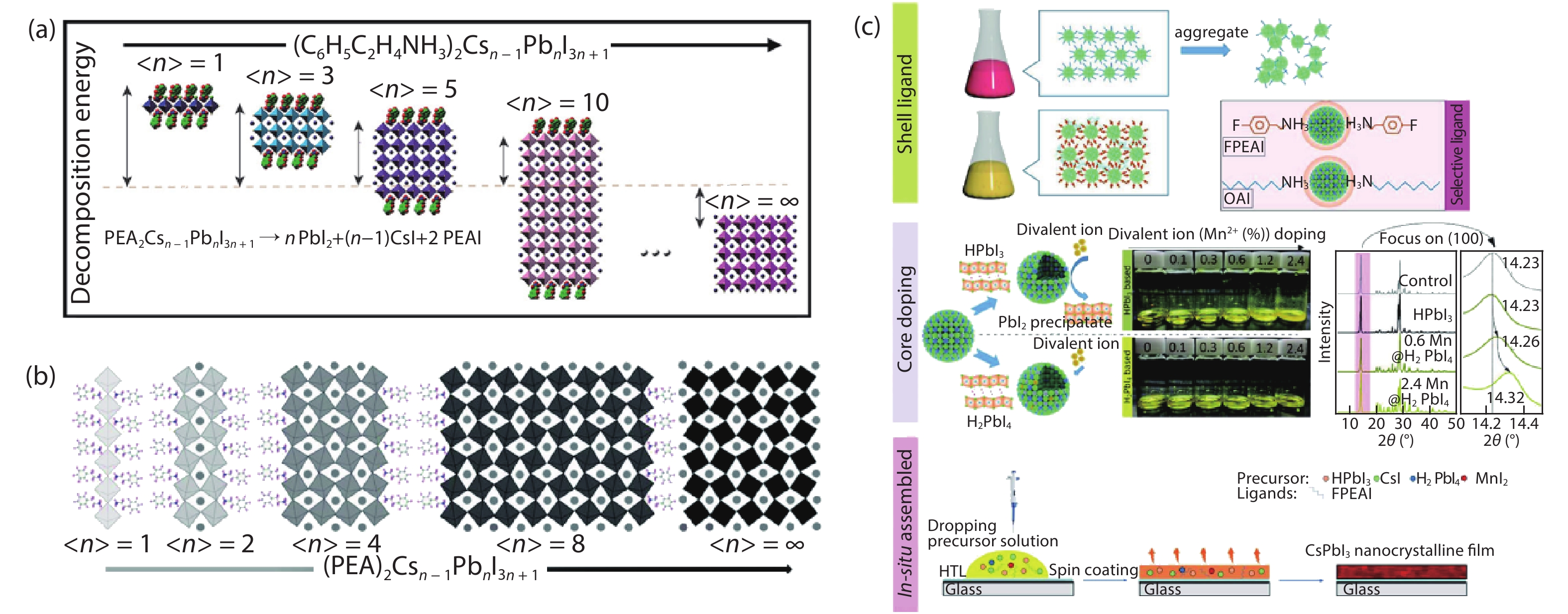
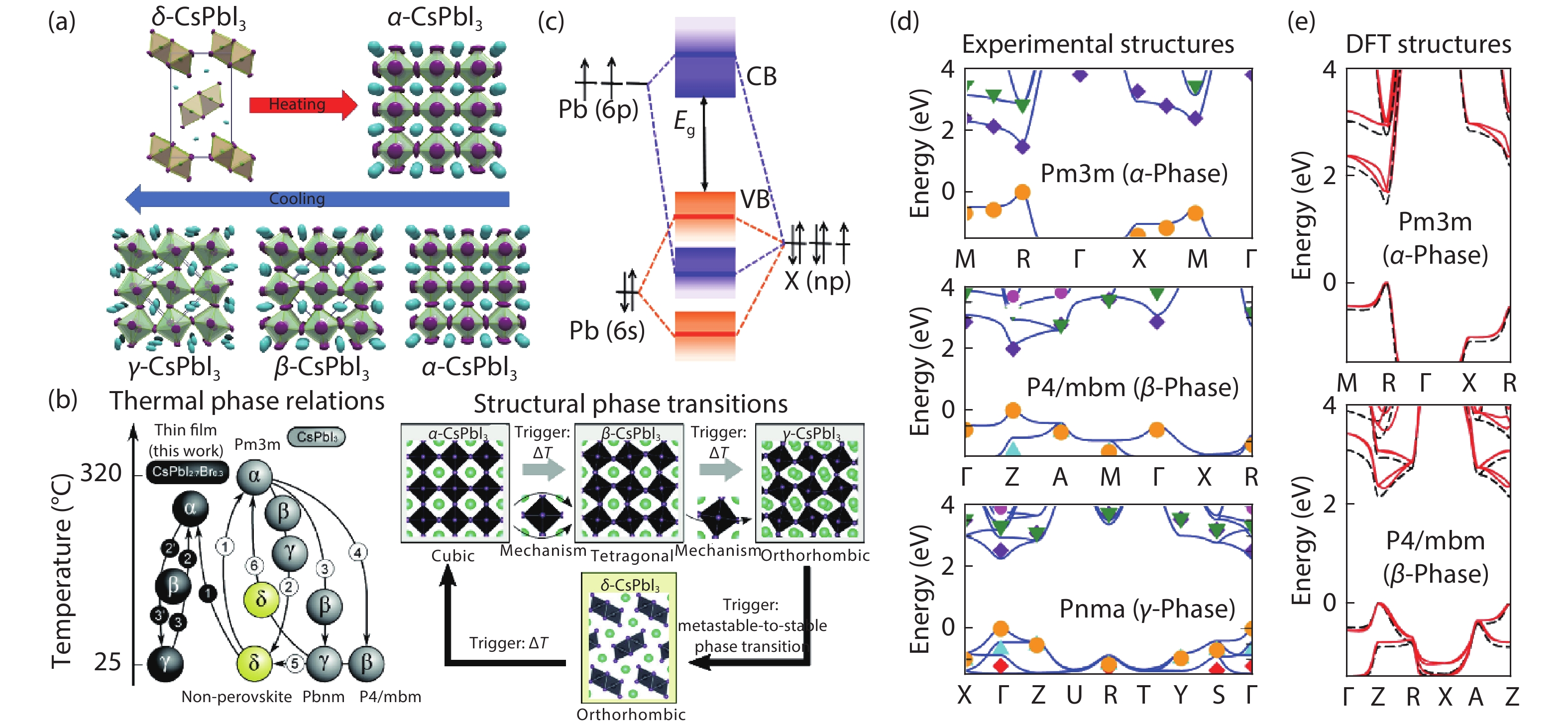
[1] Kojima A, Teshima K, Shirai Y, et al. Organometal halide perovskites as visible-light sensitizers for photovoltaic cells. J Am Chem Soc, 2009, 131, 6050 doi: 10.1021/ja809598r[2] [3] Jiang J, Wang Q, Jin Z, et al. Polymer doping for high-efficiency perovskite solar cells with improved moisture stability. Adv Energy Mater, 2018, 8, 1701757 doi: 10.1002/aenm.201701757[4] Jiang J, Jin Z, Gao F, et al. CsPbCl3-driven low-trap-density perovskite grain growth for > 20% solar cell efficiency. Adv Sci, 2018, 5, 1800474 doi: 10.1002/advs.201800474[5] Wehrenfennig C, Eperon G E, Johnston M B, et al. High charge carrier mobilities and lifetimes in organolead trihalide perovskites. Adv Mater, 2014, 26, 1584 doi: 10.1002/adma.201305172[6] Hu W, Cong H, Huang W, et al. Germanium/perovskite heterostructure for high-performance and broadband photodetector from visible to infrared telecommunication band. Light: Sci Appl, 2019, 8, 106 doi: 10.1038/s41377-019-0218-y[7] D'Innocenzo V, Grancini G, Alcocer M J P, et al. Excitons versus free charges in organo-lead tri-halide perovskites. Nat Commun, 2014, 5, 3586 doi: 10.1038/ncomms4586[8] Lin Q, Armin A, Nagiri R C R, et al. Electro-optics of perovskite solar cells. Nat Photon, 2014, 9, 106 doi: 10.1038/nphoton.2014.284[9] Fang H H, Wang F, Adjokatse S, et al. Photoexcitation dynamics in solution-processed formamidinium lead iodide perovskite thin films for solar cell applications. Light: Sci Appl, 2016, 5, e16056 doi: 10.1038/lsa.2016.56[10] Noh J H, Im S H, Heo J H, et al. Chemical management for colorful, efficient, and stable inorganic–organic hybrid nanostructured solar cells. Nano Lett, 2013, 13, 1764 doi: 10.1021/nl400349b[11] Bian H, Bai D, Jin Z, et al. Graded bandgap CsPbI2+ xBr1– x perovskite solar cells with a stabilized efficiency of 14.4%. Joule, 2018, 2, 1500 doi: 10.1016/j.joule.2018.04.012[12] Stranks S D, Eperon G E, Grancini G, et al. Electron-hole diffusion lengths exceeding 1 micrometer in an organometal trihalide perovskite absorber. Science, 2013, 342, 341 doi: 10.1126/science.1243982[13] Wang H, Bian H, Jin Z, et al. Synergy of hydrophobic surface capping and lattice contraction for stable and high-efficiency inorganic CsPbI2Br perovskite solar cells. Solar RRL, 2018, 2, 1800216 doi: 10.1002/solr.201800216[14] Stoumpos C C, Malliakas C D, Kanatzidis M G. Semiconducting tin and lead iodide perovskites with organic cations: phase transitions, high mobilities, and near-infrared photoluminescent properties. Inorg Chem, 2013, 52, 9019 doi: 10.1021/ic401215x[15] Zhao Y C, Zhou W K, Zhou X, et al. Quantification of light-enhanced ionic transport in lead iodide perovskite thin films and its solar cell applications. Light: Sci Appl, 2017, 6, e16243 doi: 10.1038/lsa.2016.243[16] Xiao C, Li Z, Guthrey H, et al. Mechanisms of electron-beam-induced damage in perovskite thin films revealed by cathodoluminescence spectroscopy. J Phys Chem C, 2015, 119, 26904 doi: 10.1021/acs.jpcc.5b09698[17] Akbulatov A F, Luchkin S Y, Frolova L A, et al. Probing the intrinsic thermal and photochemical stability of hybrid and inorganic lead halide perovskites. J Phys Chem Lett, 2017, 8, 1211 doi: 10.1021/acs.jpclett.6b03026[18] Zhou W, Zhao Y, Zhou X, et al. Light-independent ionic transport in inorganic perovskite and ultrastable cs-based perovskite solar cells. J Phys Chem Lett, 2017, 8, 4122 doi: 10.1021/acs.jpclett.7b01851[19] Wang Q, Zhang X, Jin Z, et al. Energy-down-shift CsPbCl3:Mn quantum dots for boosting the efficiency and stability of perovskite solar cells. ACS Energy Lett, 2017, 2, 1479 doi: 10.1021/acsenergylett.7b00375[20] Jin Z, Yan J, Huang X, et al. Solution-processed transparent coordination polymer electrode for photovoltaic solar cells. Nano Energy, 2017, 40, 376 doi: 10.1016/j.nanoen.2017.08.028[21] Jiang J, Jin Z, Lei J, et al. ITIC surface modification to achieve synergistic electron transport layer enhancement for planar-type perovskite solar cells with efficiency exceeding 20%. J Mater Chem A, 2017, 5, 9514 doi: 10.1039/C7TA01636K[22] Beal R E, Slotcavage D J, Leijtens T, et al. Cesium lead halide perovskites with improved stability for tandem solar cells. J Phys Chem Lett, 2016, 7, 746 doi: 10.1021/acs.jpclett.6b00002[23] Jia X, Zuo C, Tao S, et al. CsPb(I xBr1− x)3 solar cells. Sci Bull, 2019, 64, 1532 doi: 10.1016/j.scib.2019.08.017[24] Zhang X, Jin Z, Zhang J, et al. All-ambient processed binary CsPbBr3-CsPb2Br5 perovskites with synergistic enhancement for high-efficiency Cs-Pb-Br-based solar cells. ACS Appl Mater Interfaces, 2018, 10, 7145 doi: 10.1021/acsami.7b18902[25] Zhang J, Bai D, Jin Z, et al. 3D–2D–0D interface profiling for record efficiency all-inorganic CsPbBrI2 perovskite solar cells with superior stability. Adv Energy Mater, 2018, 8, 1703246 doi: 10.1002/aenm.201703246[26] Bai D, Zhang J, Jin Z, et al. Interstitial Mn2+-driven high-aspect-ratio grain growth for low-trap-density microcrystalline films for record efficiency CsPbI2Br solar cells. ACS Energy Lett, 2018, 3, 970 doi: 10.1021/acsenergylett.8b00270[27] Zhang Y Y, Chen S, Xu P, et al. Intrinsic instability of the hybrid halide perovskite semiconductor CH3NH3PbI3. Chin Phys Lett, 2018, 35, 036104 doi: 10.1088/0256-307X/35/3/036104[28] Kang C H, Dursun I, Liu G, et al. High-speed colour-converting photodetector with all-inorganic CsPbBr3 perovskite nanocrystals for ultraviolet light communication. Light: Sci Appl, 2019, 8, 94 doi: 10.1038/s41377-019-0204-4[29] Liu G, Zhou C, Wan F, et al. Dependence of power conversion properties of perovskite solar cells on operating temperature. Appl Phys Lett, 2018, 113, 3501 doi: 10.1063/1.5041028[30] Liu G, Yang B, Liu B, et al. Irreversible light-soaking effect of perovskite solar cells caused by light-induced oxygen vacancies in titanium oxide. Appl Phys Lett, 2017, 111, 3501 doi: 10.1063/1.4994085[31] Wang J F, Lin D X, Yuan Y B. Recent progress of ion migration in organometal halide perovskite. Acta Phys Sin, 2019, 68, 158801 doi: 10.7498/aps.68.20190853[32] Ahmad W, Khan J, Niu G, et al. Inorganic CsPbI3 perovskite-based solar cells: a choice for a tandem device. Solar RRL, 2017, 1, 1700048 doi: 10.1002/solr.201700048[33] Wang P, Zhang X, Zhou Y, et al. Solvent-controlled growth of inorganic perovskite films in dry environment for efficient and stable solar cells. Nat Commun, 2018, 9, 2225 doi: 10.1038/s41467-018-04636-4[34] Zhang X, Wang Q, Jin Z, et al. Stable ultra-fast broad-bandwidth photodetectors based on α-CsPbI3 perovskite and NaYF4:Yb,Er quantum dots. Nanoscale, 2017, 9, 6278 doi: 10.1039/C7NR02010D[35] Steele J A, Jin H D, Iurii I, et al. Thermal unequilibrium of strained black CsPbI3 thin films. Science, 2019, 365, 679 doi: 10.1126/science.aax3878[36] Hoffman J B, Schleper A L, Kamat P V. Transformation of sintered CsPbBr3 nanocrystals to cubic CsPbI3 and gradient CsPbBr xI3– x through halide exchange. J Am Chem Soc, 2016, 138, 8603 doi: 10.1021/jacs.6b04661[37] Wang Q, Jin Z, Chen D, et al. μ-graphene crosslinked CsPbI3 quantum dots for high efficiency solar cells with much improved stability. Adv Energy Mater, 2018, 8, 1800007 doi: 10.1002/aenm.201800007[38] Zhao H, Xu J, Zhou S, et al. Preparation of tortuous 3D γ-CsPbI3 films at low temperature by CaI2 as dopant for highly efficient perovskite solar cells. Adv Funct Mater, 2019, 29, 1808986 doi: 10.1002/adfm.201808986[39] Dayan A S, Cohen B E, Aharon S, et al. Enhancing stability and photostability of CsPbI3 by reducing its dimensionality. Chem Mater, 2018, 30, 8017 doi: 10.1021/acs.chemmater.8b03709[40] Ye T, Zhou B, Zhan F, et al. Below 200 °C fabrication strategy of black phase CsPbI3 film for ambient-air-stable solar cells. Solar RRL, 2019, 10 doi: 10.1002/solr.202000014[41] Xiang S, Li W, Wei Y, et al. Natrium doping pushes the efficiency of carbon-based CsPbI3 perovskite solar cells to 10.7%. iScience, 2019, 15, 156 doi: 10.1016/j.isci.2019.04.025[42] Eperon G E, Paternò G M, Sutton R J, et al. Inorganic caesium lead iodide perovskite solar cells. J Mater Chem A, 2015, 3, 19688 doi: 10.1039/C5TA06398A[43] Wang K, Jin Z, Liang L, et al. All-inorganic cesium lead iodide perovskite solar cells with stabilized efficiency beyond 15%. Nat Commun, 2018, 9, 4544 doi: 10.1038/s41467-018-06915-6[44] Wang Y, Zhang T, Kan M, et al. Bifunctional stabilization of all-inorganic α-CsPbI3 perovskite for 17% efficiency photovoltaics. J Am Chem Soc, 2018, 140, 12345 doi: 10.1021/jacs.8b07927[45] Swarnkar A, Ravi V K, Nag A. Beyond colloidal cesium lead halide perovskite nanocrystals: analogous metal halides and doping. ACS Energy Lett, 2017, 2, 1089 doi: 10.1021/acsenergylett.7b00191[46] Marronnier A, Roma G, Boyer-Richard S, et al. Anharmonicity and disorder in the black phases of cesium lead iodide used for stable inorganic perovskite solar cells. ACS Nano, 2018, 12, 3477 doi: 10.1021/acsnano.8b00267[47] Bai D, Bian H, Jin Z, et al. Temperature-assisted crystallization for inorganic CsPbI2Br perovskite solar cells to attain high stabilized efficiency 14.81%. Nano Energy, 2018, 52, 408 doi: 10.1016/j.nanoen.2018.08.012[48] Green M A, Ho-Baillie A, Snaith H J. The emergence of perovskite solar cells. Nat Photon, 2014, 8, 506 doi: 10.1038/nphoton.2014.134[49] Eperon G E, Stranks S D, Menelaou C, et al. Formamidinium lead trihalide: a broadly tunable perovskite for efficient planar heterojunction solar cells. Energy Environ Sci, 2014, 7, 982 doi: 10.1039/c3ee43822h[50] Zhang J, Hodes G, Jin Z, et al. All-inorganic CsPbX3 perovskite solar cells: progress and prospects. Angew Chem Int Ed, 2019, 58, 15596 doi: 10.1002/anie.201901081[51] Huang Y, Yin W J, He Y. Intrinsic point defects in inorganic cesium lead iodide perovskite CsPbI3. J Phys Chem C, 2018, 122, 1345 doi: 10.1021/acs.jpcc.7b10045[52] Sutton R J, Filip M R, Haghighirad A A, et al. Cubic or orthorhombic? revealing the crystal structure of metastable black-phase CsPbI3 by theory and experiment ACS Energy Lett, 2018, 3, 1787 doi: 10.1021/acsenergylett.8b00672[53] Sun J K, Huang S, Liu X Z, et al. Polar solvent induced lattice distortion of cubic CsPbI3 nanocubes and hierarchical self-assembly into orthorhombic single-crystalline nanowires. J Am Chem Soc, 2018, 140, 11705 doi: 10.1021/jacs.8b05949[54] Ravi V K, Markad G B, Nag A. Band edge energies and excitonic transition probabilities of colloidal CsPbX3 (X = Cl, Br, I) perovskite nanocrystals. ACS Energy Lett, 2016, 1, 665 doi: 10.1021/acsenergylett.6b00337[55] Stoumpos C C, Kanatzidis M G. The renaissance of halide perovskites and their evolution as emerging semiconductors. Acc Chem Res, 2015, 48, 2791 doi: 10.1021/acs.accounts.5b00229[56] Katan C, Pedesseau L, Kepenekian M, et al. Interplay of spin–orbit coupling and lattice distortion in metal substituted 3D tri-chloride hybrid perovskites. J Mater Chem A, 2015, 3, 9232 doi: 10.1039/C4TA06418F[57] Zheng L, Zhang D, Ma Y, et al. Morphology control of the perovskite films for efficient solar cells. Dalton Trans, 2015, 44, 10582 doi: 10.1039/C4DT03869J[58] Soe C M M, Stoumpos C C, Harutyunyan B, et al. Room temperature phase transition in methylammonium lead iodide perovskite thin films induced by hydrohalic acid additives. ChemSusChem, 2016, 9, 2656 doi: 10.1002/cssc.201600879[59] Sharenko A, Mackeen C, Jewell L, et al. Evolution of iodoplumbate complexes in methylammonium lead iodide perovskite precursor solutions. Chem Mater, 2017, 29, 1315 doi: 10.1021/acs.chemmater.6b04917[60] Mohamad D K, Freestone B G, Masters R, et al. Optimized organometal halide perovskite solar cell fabrication through control of nanoparticle crystal patterning. J Mater Chem C, 2017, 5, 2352 doi: 10.1039/C6TC05189H[61] Haque F, Wright M, Mahmud M A, et al. Effects of hydroiodic acid concentration on the properties of CsPbI3 perovskite solar cells. ACS Omega, 2018, 3, 11937 doi: 10.1021/acsomega.8b01589[62] Kim Y G, Kim T Y, Oh J H, et al. cesium lead iodide solar cells controlled by annealing temperature. Phys Chem Chem Phys, 2017, 19, 6257 doi: 10.1039/C6CP08177K[63] Heo D Y, Han S M, Woo N S, et al. Role of additives on the performance of CsPbI3 solar cells. J Phys Chem C, 2018, 122, 15903 doi: 10.1021/acs.jpcc.8b04613[64] Wei Y, Li W, Xiang S, et al. Precursor effects on methylamine gas-induced CH3NH3PbI3 films for stable carbon-based perovskite solar cells. Solar Energy, 2018, 174, 139 doi: 10.1016/j.solener.2018.09.003[65] Wang F, Yu H, Xu H, et al. HPbI3: a new precursor compound for highly efficient solution-processed perovskite solar cells. Adv Funct Mater, 2015, 25, 1120 doi: 10.1002/adfm.201404007[66] Pang S, Zhou Y, Wang Z, et al. Transformative evolution of organolead triiodide perovskite thin films from strong room-temperature solid-gas interaction between HPbI3–CH3NH2 precursor pair. J Am Chem Soc, 2016, 138, 750 doi: 10.1021/jacs.5b11824[67] Ding X, Chen H, Wu Y, et al. Triple cation additive NH3+C2H4NH2+C2H4NH3+-induced phase-stable inorganic α-CsPbI3 perovskite films for use in solar cells. J Mater Chem A, 2018, 6, 18258 doi: 10.1039/C8TA04590A[68] Xiang S, Fu Z, Li W, et al. Highly air-stable carbon-based α-CsPbI3 perovskite solar cells with a broadened optical spectrum. ACS Energy Lett, 2018, 3, 1824 doi: 10.1021/acsenergylett.8b00820[69] Wang Y, Zhang T, Xu F, et al. A Facile low temperature fabrication of high performance CsPbI2Br all-inorganic perovskite solar cells. Solar RRL, 2018, 2, 1700180 doi: 10.1002/solr.201700180[70] Ye Q, Zhao Y, Mu S, et al. Cesium lead inorganic solar cell with efficiency beyond 18% via reduced charge recombination. Adv Mater, 2019, 31, e1905143 doi: 10.1002/adma.201905143[71] Ke W, Spanopoulos I, Stoumpos C C, et al. Myths and reality of HPbI3 in halide perovskite solar cells. Nat Commun, 2018, 9, 4785 doi: 10.1038/s41467-018-07204-y[72] Pei Y, Liu Y, Li F, et al. Unveiling property of hydrolysis-derived DMAPbI3 for perovskite devices: composition engineering, defect mitigation, and stability optimization. iScience, 2019, 15, 165 doi: 10.1016/j.isci.2019.04.024[73] Wang Y, Dar M I, Ono L K, et al. Thermodynamically stabilized β-CsPbI3–based perovskite solar cells with efficiencies >18%. Science, 2019, 365, 591 doi: 10.1126/science.aav8680[74] Wang Y, Liu X, Zhang T, et al. the role of dimethylammonium iodide in CsPbI3 perovskite fabrication: additive or dopant. Angew Chem Int Ed, 2019, 58, 16691 doi: 10.1002/anie.201910800[75] Meng H, Shao Z, Wang L, et al. Chemical composition and phase evolution in DMAI-derived inorganic perovskite solar cells. ACS Energy Lett, 2020, 5, 263 doi: 10.1021/acsenergylett.9b02272[76] Bian H, Wang H, Li Z, et al. Unveiling the effects of hydrolysis-derived DMAI/DMAPbI x intermediate compound on performance of CsPbI3 solar cells. Adv Sci, 2019, 10, 1902868 doi: 10.1002/advs.201902868[77] Dutta A, Pradhan N. Phase-stable red-emitting CsPbI3 nanocrystals: successes and challenges. ACS Energy Lett, 2019, 4, 709 doi: 10.1021/acsenergylett.9b00138[78] Zhang T, Dar M I, Li G, et al. Bication lead iodide 2D perovskite component to stabilize inorganic α-CsPbI3 perovskite phase for high-efficiency solar cells. Adv Sci, 2017, 3, e1700841 doi: 10.1126/sciadv.1700841[79] Wang Y, Zhang T, Kan M, et al. Efficient α-CsPbI3 photovoltaics with surface terminated organic cations. Joule, 2018, 2, 2065 doi: 10.1016/j.joule.2018.06.013[80] Wu T, Wang Y, Dai Z, et al. Efficient and stable CsPbI3 solar cells via regulating lattice distortion with surface organic terminal groups. Adv Mater, 2019, 31, e1900605 doi: 10.1002/adma.201900605[81] Fu Y, Rea M T, Chen J, et al. Selective stabilization and photophysical properties of metastable perovskite polymorphs of CsPbI3 in thin films. Chem Mater, 2017, 29, 8385 doi: 10.1021/acs.chemmater.7b02948[82] Becker P, Márquez J A, Just J, et al. Low temperature synthesis of stable γ-CsPbI3 perovskite layers for solar cells obtained by high throughput experimentation. Adv Energy Mater, 2019, 9, 1900555 doi: 10.1002/aenm.201900555[83] Zhao B, Jin S, Huang S, et al. Thermodynamically stable orthorhombic γ-CsPbI3 thin films for high-performance photovoltaics. J Am Chem Soc, 2018, 140, 11716 doi: 10.1021/jacs.8b06050[84] Liu C, Yang Y, Xia X, et al. Soft Template-controlled growth of high-quality CsPbI3 films for efficient and stable solar cells. Adv Energy Mater, 2020, 10, 1903751 doi: 10.1002/aenm.201903751[85] Liang L, Zhizai L, Zhou F, et al. Humidity-insensitive fabrication of efficient CsPbI3 solar cells in ambient air. J Mater Chem A, 2019, 7, 26776 doi: 10.1039/C9TA10597B[86] Wang H, Bian H, Jin Z, et al. Cesium lead mixed-halide perovskites for low-energy loss solar cells with efficiency beyond 17%. Chem Mater, 2019, 31, 6231 doi: 10.1021/acs.chemmater.9b02248[87] Wang K, Jin Z, Liang L, et al. Chlorine doping for black γ-CsPbI3 solar cells with stabilized efficiency beyond 16%. Nano Energy, 2019, 58, 175 doi: 10.1016/j.nanoen.2019.01.034[88] Yao Z, Jin Z, Zhang X, et al. Pseudohalide (SCN-)-doped CsPbI3 for high performance solar cells. J Mater Chem C, 2019, 7, 13736 doi: 10.1039/C9TC04851K[89] Bian H, Wang Q, Yang S, et al. Nitrogen-doped graphene quantum dots for 80% photoluminescence quantum yield for inorganic γ-CsPbI3 perovskite solar cells with efficiency beyond 16%. J Mater Chem A, 2019, 7, 5740 doi: 10.1039/C8TA12519H[90] Liang L, Liu M, Jin Z, et al. Optical Management with nanoparticles for a light conversion efficiency enhancement in inorganic γ-CsPbI3 solar cells. Nano Lett, 2019, 19, 1796 doi: 10.1021/acs.nanolett.8b04842[91] Bian H, Wang Q, Ding L, et al. Light management via tuning the fluorine-doped tin oxide glass haze-drives high-efficiency CsPbI3 solar cells. Phys Status Solidi A, 2019, 216, 1900602 doi: 10.1002/pssa.201900602[92] Wang Q, Zheng X, Deng Y, et al. Stabilizing the α-phase of CsPbI3 perovskite by sulfobetaine zwitterions in one-step spin-coating films. Joule, 2017, 1, 371 doi: 10.1016/j.joule.2017.07.017[93] Jin Z, Yuan M, Li H, et al. Graphdiyne: an efficient hole transporter for stable high-performance colloidal quantum dot solar cells. Adv Funct Mater, 2016, 26, 5284 doi: 10.1002/adfm.201601570[94] Jin Z, Wang A, Zhou Q, et al. Detecting trap states in planar PbS colloidal quantum dot solar cells. Sci Rep, 2016, 6, 37106 doi: 10.1038/srep37106[95] Yao H, Zhou F, Li Z, et al. Strategies for improving the stability of tin-based perovskite (ASnX3) solar cells. Adv Sci, 2020, 10, 1903540 doi: 10.1002/advs.201903540[96] Jin Z, Zhou Q, Chen Y, et al. Graphdiyne:ZnO nanocomposites for high-performance UV photodetectors. Adv Mater, 2016, 28, 3697 doi: 10.1002/adma.201600354[97] Jiang Y, Yuan J, Ni Y, et al. Reduced-dimensional α-CsPbX3 perovskites for efficient and stable photovoltaics. Joule, 2018, 2, 1356 doi: 10.1016/j.joule.2018.05.004[98] Wang K, Li Z, Zhou F, et al. Ruddlesden–popper 2D component to stabilize γ-CsPbI3 Perovskite phase for stable and efficient photovoltaics. Adv Energy Mater, 2019, 9, 1902529 doi: 10.1002/aenm.201902529[99] Wang H, Xiang S, Li W, et al. Skillfully deflecting the question: a small amount of piperazine-1,4-diium iodide radically enhances the thermal stability of CsPbI3 perovskite. J Mater Chem C, 2019, 7, 11757 doi: 10.1039/C9TC03576A[100] Dutta A, Dutta S K, Das Adhikari S, et al. Phase-stable CsPbI3 nanocrystals: the reaction temperature matters. Angew Chem Int Ed, 2018, 57, 9083 doi: 10.1002/anie.201803701[101] Xi J, Piao C, Byeon J, et al. Rational core-shell design of open air low temperature in situ processable CsPbI3 quasi-nanocrystals for stabilized p –i –n solar cells. Adv Energy Mater, 2019, 9, 1901787 doi: 10.1002/aenm.201901787[102] Li B, Zhang Y, Fu L, et al. Surface passivation engineering strategy to fully-inorganic cubic CsPbI3 perovskites for high-performance solar cells. Nat Commun, 2018, 9, 1076 doi: 10.1038/s41467-018-03169-0[103] Gan J, He J, Hoye R L Z, et al. α-CsPbI3 colloidal quantum dots: synthesis, photodynamics, and photovoltaic applications. ACS Energy Lett, 2019, 4, 1308 doi: 10.1021/acsenergylett.9b00634[104] Zhang X, Munir R, Xu Z, et al. Phase transition control for high performance ruddlesden-popper perovskite solar cells. Adv Mater, 2018, 30, 1707166 doi: 10.1002/adma.201707166 -
Proportional views





 DownLoad:
DownLoad: