| Citation: |
L. Castañeda. Transparent conductive stannic oxide coatings employing an ultrasonic spray pyrolysis technique: The relevance of the molarity content in the aerosol solution for improvement the electrical properties[J]. Journal of Semiconductors, 2022, 43(2): 022802. doi: 10.1088/1674-4926/43/2/022802
****
L. Castañeda, Transparent conductive stannic oxide coatings employing an ultrasonic spray pyrolysis technique: The relevance of the molarity content in the aerosol solution for improvement the electrical properties[J]. J. Semicond., 2022, 43(2): 022802. doi: 10.1088/1674-4926/43/2/022802.
|
Transparent conductive stannic oxide coatings employing an ultrasonic spray pyrolysis technique: The relevance of the molarity content in the aerosol solution for improvement the electrical properties
DOI: 10.1088/1674-4926/43/2/022802
More Information
-
Abstract
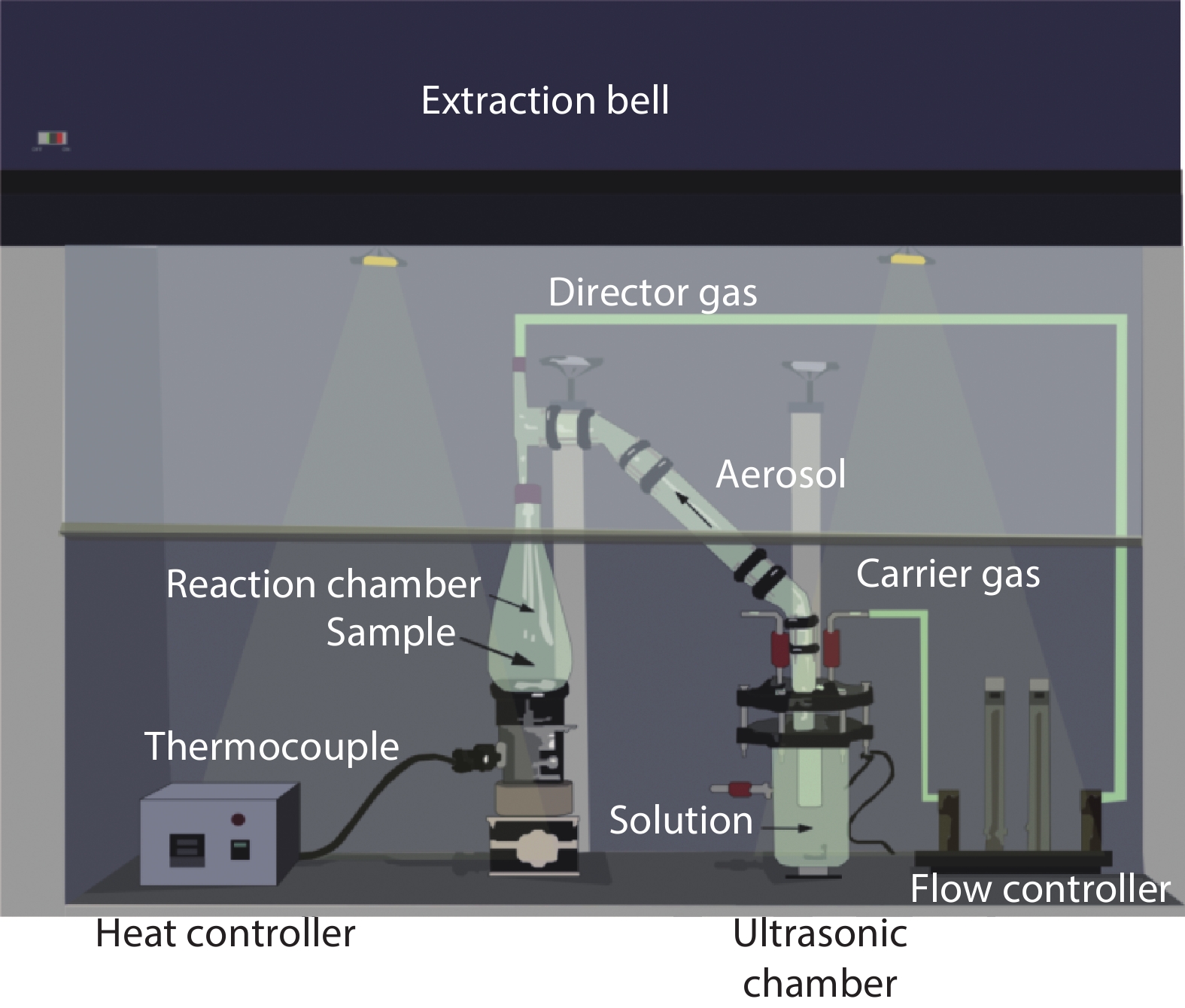
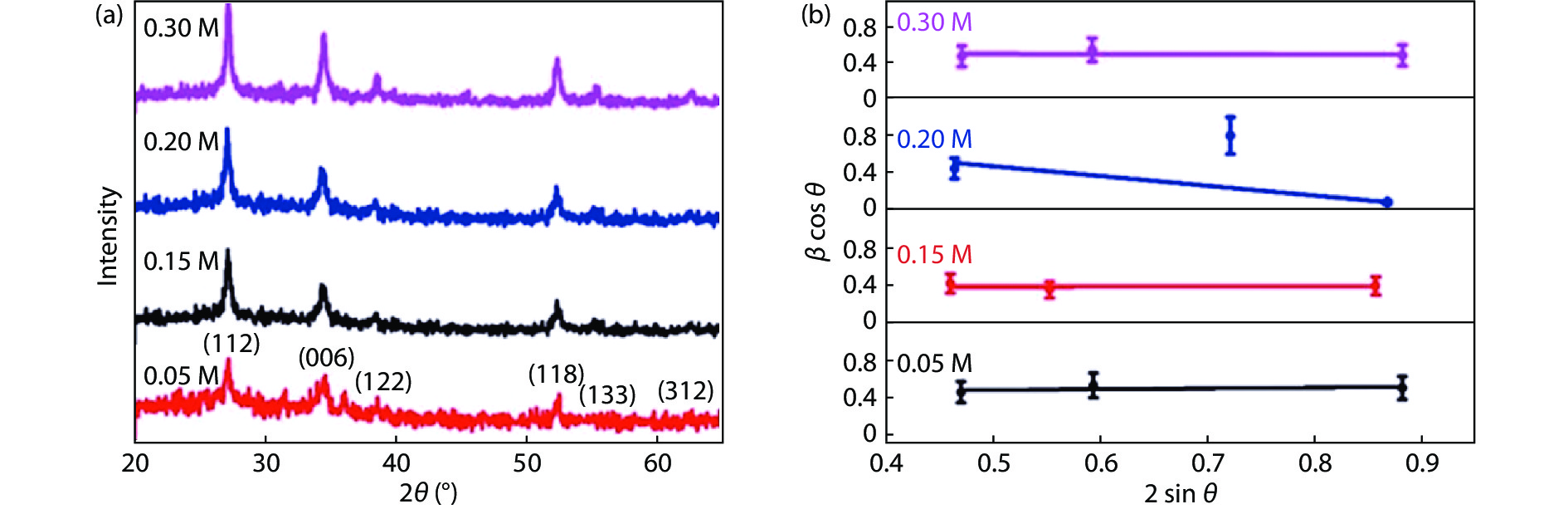
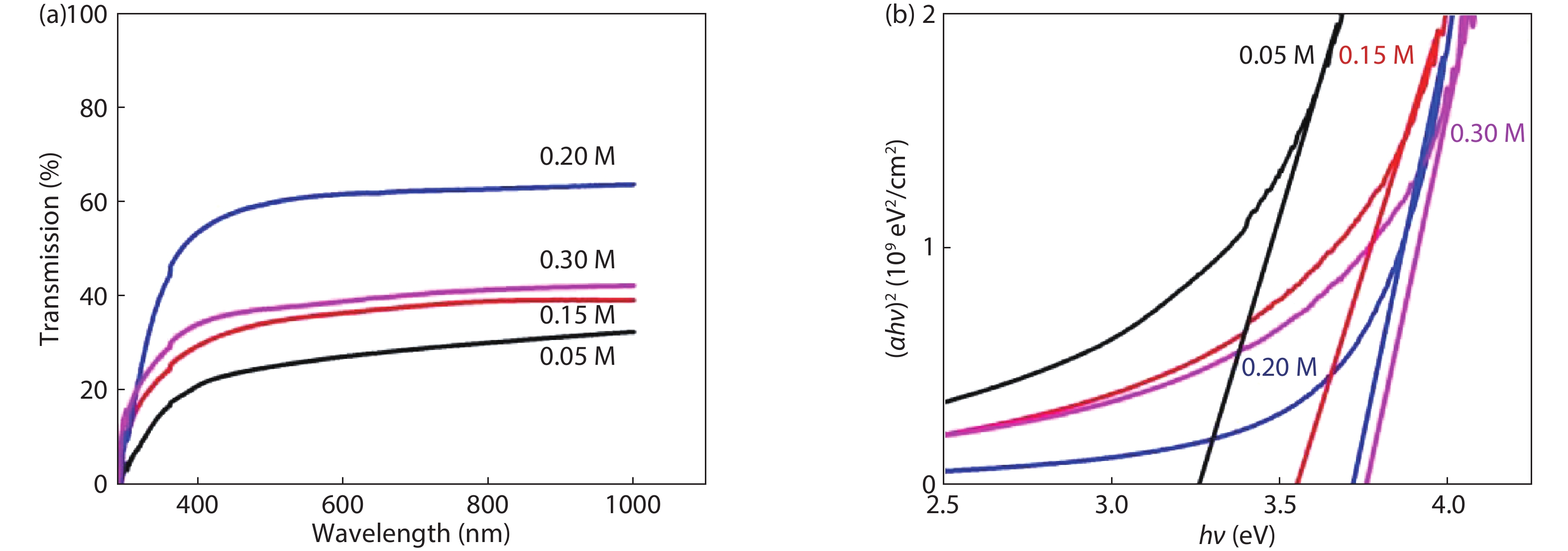
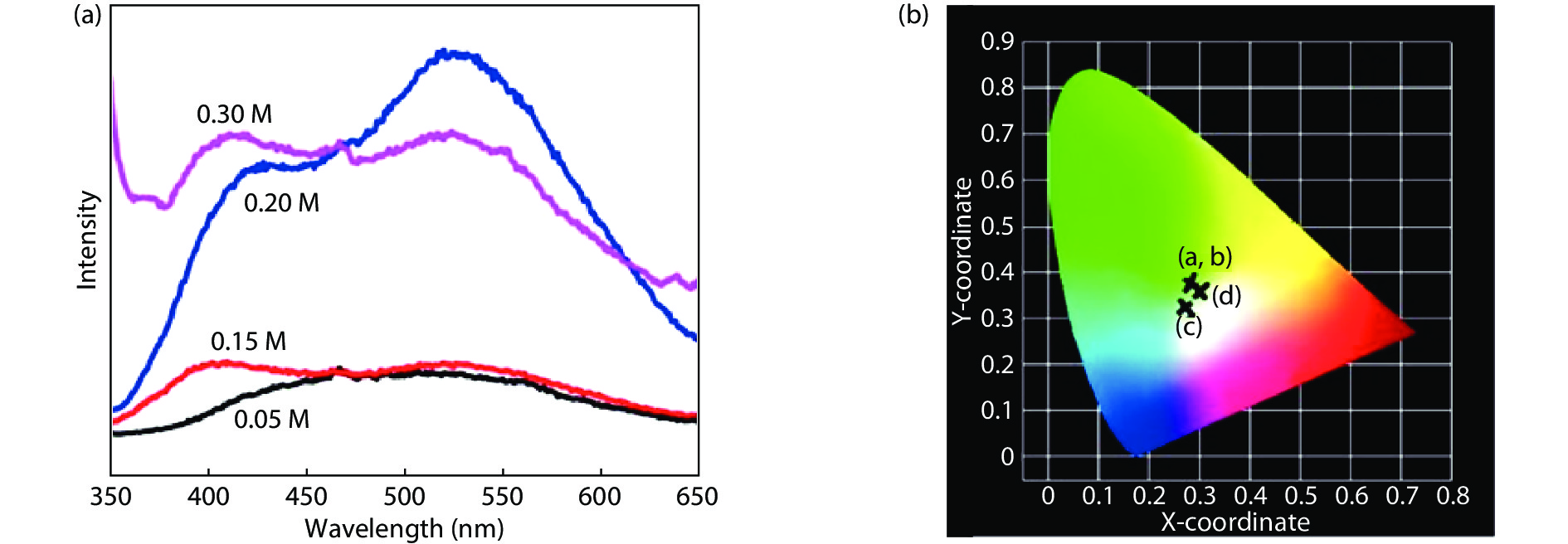
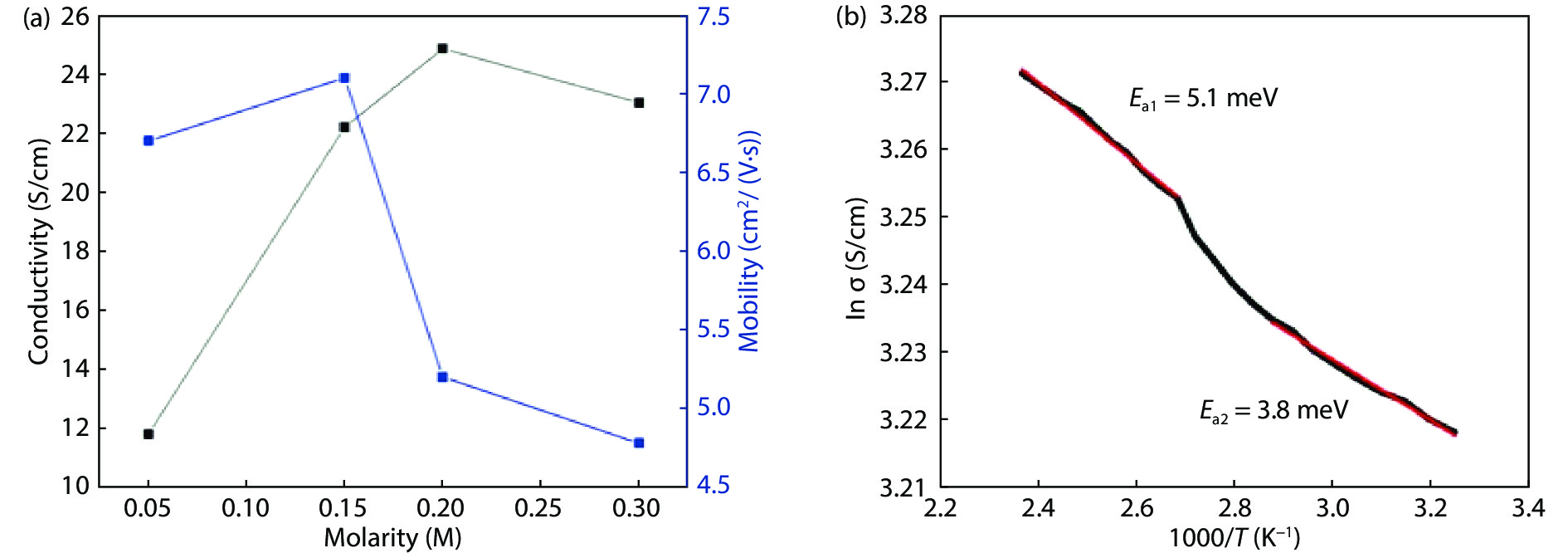
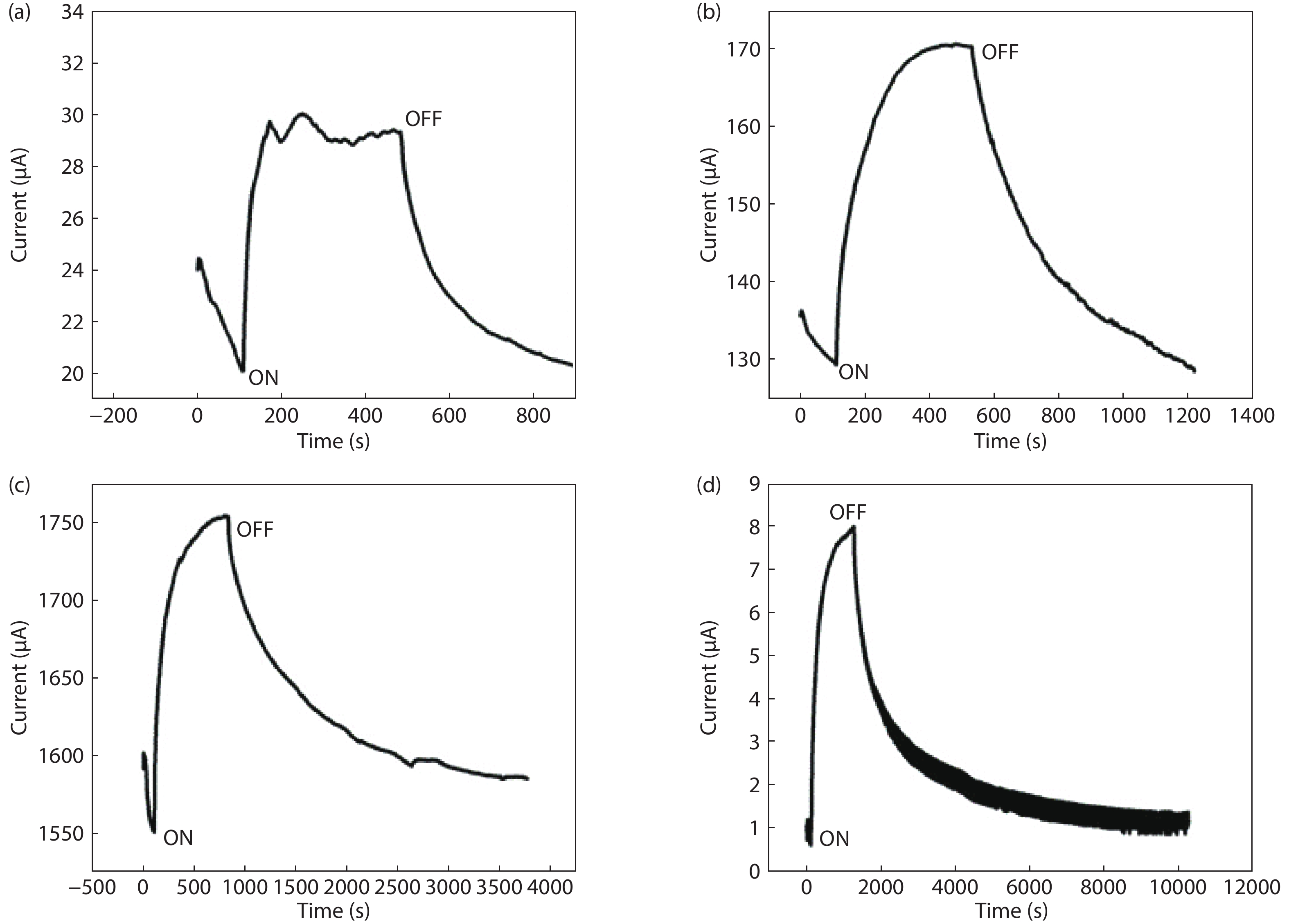
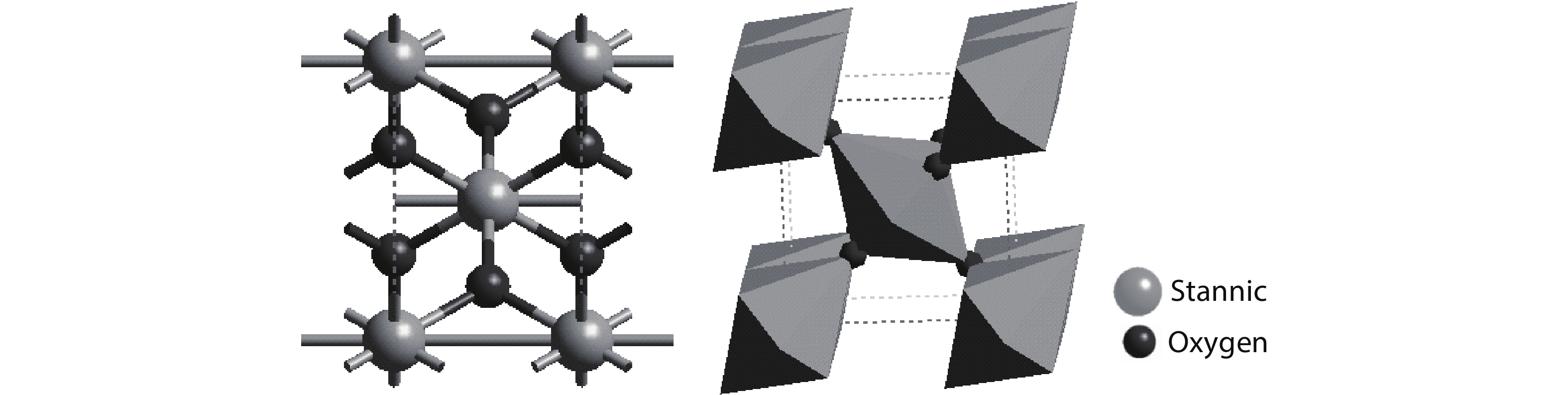
Highly transparent conductive stoichiometric nanocrystalline stannic oxide coatings were deposited onto Corning® EAGLE XG® slim glass substrates. Including each coating, it was deposited for various concentrations in the aerosol solution with the substrate temperature maintained at 623.15 K by an ultrasonic spray pyrolysis (USP) technique. Nitrogen was employed both as the solution carrier in addition to aerosol directing gas, maintaining its flow rates at 3500.0 and 500.0 mL/min, respectively. The coatings were polycrystalline, with preferential growth along the stannic oxide (112) plane, irrespective of the molarity content in the spray solution. The coating prepared at 0.2 M, a concentration in the aerosol solution, showed an average transmission of 60% in the visible light region spectrum with a maximum conductivity of 24.86 S/cm. The coatings deposited exhibited in the general photoluminescence spectrum emission colors of green, greenish white, and bluish white calculated on the intensities of the excitonic and oxygen vacancy defect level emissions.-
Keywords:
- stannic oxide,
- coatings,
- ultrasonic spray pyrolysis
-
References
[1] Castañeda L. Present status of the development and application of transparent conductors oxide thin solid films. Mater Sci Appl, 2011, 2, 1233 doi: 10.4236/msa.2011.29167[2] Yuliarto. Study of porous nanoflake ZnO for dye-sensitized solar cell application. Am J Eng Appl Sci, 2009, 2, 236 doi: 10.3844/ajeassp.2009.236.240[3] Castañeda L, Avendaño-Alejo M. Physical characterization of nickel-doped zinc oxide thin solid films deposited by the ultrasonic chemically sprayed technique: Gas-sensing performance in a propane atmosphere. J Nanoelectron Optoelectron, 2014, 9, 419 doi: 10.1166/jno.2014.1610[4] Castañeda L, Maldonado A, Rodríguez-Baez J, et al. Chemical spray pyrolysis deposited fluorine-doped zinc oxide thin films: Effect of acetic acid content in the starting solution on the physical properties. Mater Sci Semicond Process, 2012, 15, 232 doi: 10.1016/j.mssp.2011.06.002[5] Castañeda L, Morales-Saavedra O G, Acosta D R, et al. Structural, morphological, optical, and nonlinear optical properties of fluorine-doped zinc oxide thin films deposited on glass substrates by the chemical spray technique. Phys Status Solidi A, 2006, 203, 1971 doi: 10.1002/pssa.200521386[6] Suchea M, Christoulakis S, Moschovis K, et al. ZnO transparent thin films for gas sensor applications. Thin Solid Films, 2006, 515, 551 doi: 10.1016/j.tsf.2005.12.295[7] Castañeda L. Evaluating propane sensing properties of ultrasonic spray deposited zinc oxide thin solid films. J Nanoelectron Optoelectron, 2018, 13, 819 doi: 10.1166/jno.2018.2315[8] Hussain S, Liu T M, Aslam N, et al. Polymer-assisted co-axial multi-layered circular ZnO nanodisks. Mater Lett, 2015, 152, 260 doi: 10.1016/j.matlet.2015.03.124[9] Hussain S, Liu T M, Kashif M, et al. A simple preparation of ZnO nanocones and exposure to formaldehyde. Mater Lett, 2014, 128, 35 doi: 10.1016/j.matlet.2014.04.115[10] Castañeda L. The effects of warmness plasma, and high frequency electrical field on beam-1 plasma interaction in plasma waveguide. J Comput Theor Nanosci, 2018, 15, 1830 doi: 10.1166/jctn.2018.7318[11] Girtan M, Rusu G G, Dabos-Seignon S, et al. Structural and electrical properties of zinc oxides thin films prepared by thermal oxidation. Appl Surf Sci, 2008, 254, 4179 doi: 10.1016/j.apsusc.2007.12.055[12] Lee J S, Islam M S, Kim S. Photoresponses of ZnO nanobridge devices fabricated using a single-step thermal evaporation method. Sens Actuators B, 2007, 126, 73 doi: 10.1016/j.snb.2006.10.042[13] Castañeda L, Avendaño-Alejo M, Gómez H, et al. Physical characterization of ruthenium-doped zinc oxide thin solid films deposited by the sol–gel technique: Gas-sensing performance in a propane atmosphere. Sen Lett, 2013, 11, 286 doi: 10.1166/sl.2013.2721[14] Castañeda L. Synthesis and characterization of ZnO micro- and nano-cages. Acta Mater, 2009, 57, 1385 doi: 10.1016/j.actamat.2008.11.022[15] Ku C H, Wu J J. Chemical bath deposition of ZnO nanowire–nanoparticle composite electrodes for use in dye-sensitized solar cells. Nanotechnology, 2007, 18, 505706 doi: 10.1088/0957-4484/18/50/505706[16] Castañeda L. Photoluminescence and morphological characterization of silver-doped zinc oxide novel nanostructures obtained by ultrasonic spray pyrolysis. J Nanoelectron Optoelectron, 2013, 8, 373 doi: 10.1166/jno.2013.1475[17] Warren B E. X-ray diffraction. New York: Dover Publications, 1990, 253[18] Castañeda L, Torres-Torres C, Trejo-Valdez M, et al. Optical and photoconductive properties exhibited by silver doped zinc oxide thin films. J Nanoelectron Optoelectron, 2013, 8, 267 doi: 10.1166/jno.2013.1465[19] Barret C. Massalki T B. Structure of metals. Oxford: Pergamon Press, 1980, 73[20] Park S. Enhancement of hydrogen sensing response of ZnO nanowires for the decoration of WO3 nanoparticles. Mater Lett, 2019, 234, 315 doi: 10.1016/j.matlet.2018.09.129[21] Aziz A, Tiwale N, Hodge S A, et al. Core-shell electrospun polycrystalline ZnO nanofibers for ultra-sensitive NO2 gas sensing. ACS Appl Mater Interfaces, 2018, 10, 43817 doi: 10.1021/acsami.8b17149[22] Jeem M, Zhang L H, Ishioka J, et al. Tuning optoelectrical properties of ZnO nanorods with excitonic defects via submerged illumination. Nano Lett, 2017, 17, 2088 doi: 10.1021/acs.nanolett.7b00324[23] Zhu R, Zhang W G, Li C, et al. Uniform zinc oxide nanowire arrays grown on nonepitaxial surface with general orientation control. Nano Lett, 2013, 13, 5171 doi: 10.1021/nl402476u[24] Zhai T Y, Li L, Ma Y, et al. One-dimensional inorganic nanostructures: Synthesis, field-emission and photodetection. Chem Soc Rev, 2011, 40, 2986 doi: 10.1039/c0cs00126k[25] Ding J N, Zhou Y R, Dong G Q, et al. Solution-processed ZnO as the efficient passivation and electron selective layer of silicon solar cells. Prog Photovolt: Res Appl, 2018, 26, 974 doi: 10.1002/pip.3044[26] Chen P L, Ma X Y, Yang D R. Ultraviolet electroluminescence from ZnO/p-Si heterojunctions. J Appl Phys, 2007, 101, 053103 doi: 10.1063/1.2464185 -
Proportional views





 DownLoad:
DownLoad: